 :max_bytes(150000):strip_icc()/PeriodicTableallcolor-58b5c82c3df78cdcd8bbb80f.png)
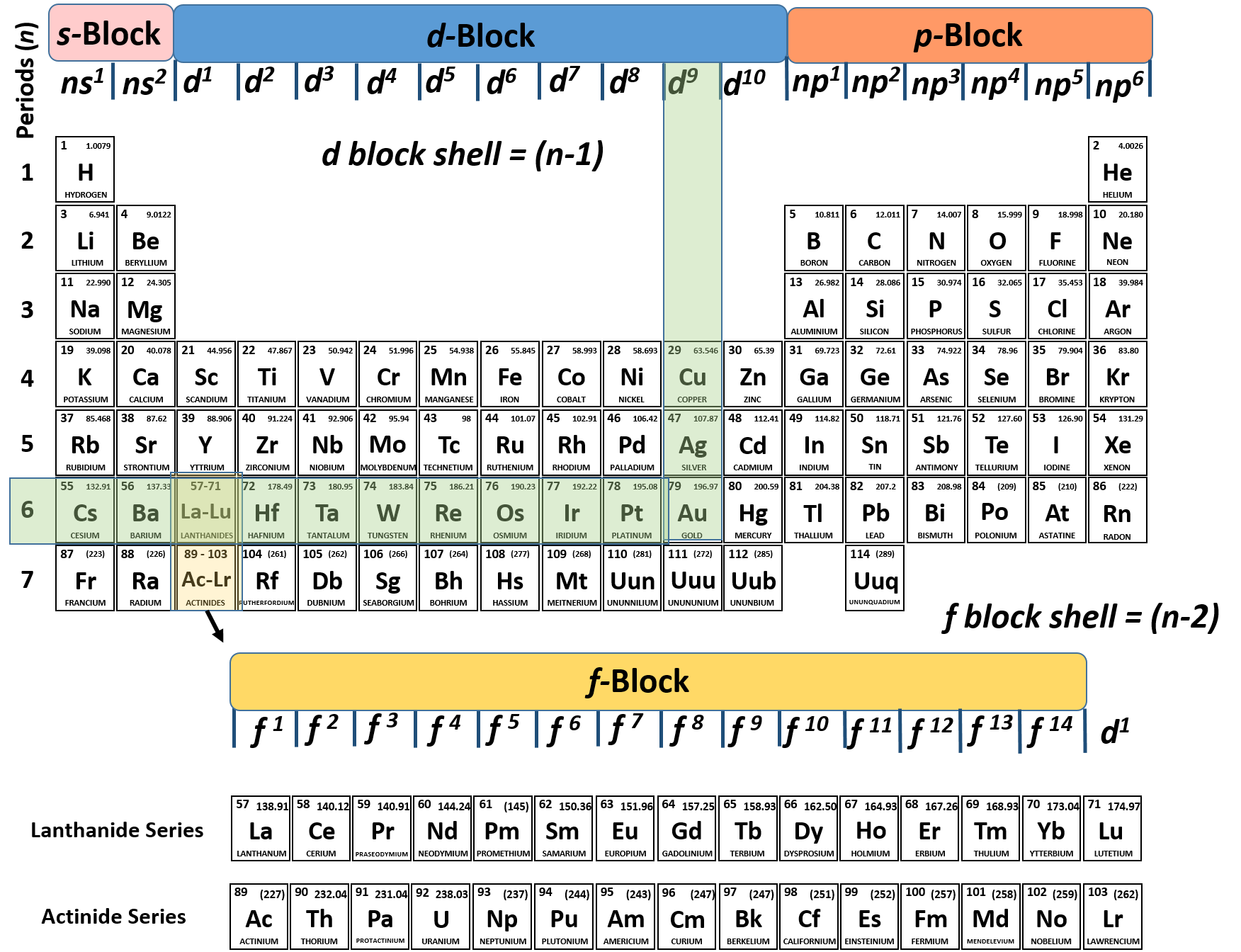
(In the modern periodic table, a group or family corresponds to one vertical column. The periodic table of chemical elements, often called the periodic table, organizes all discovered chemical elements in rows (called periods) and columns. The periodic table allows chemists a shortcut by arranging typical elements according to their properties and putting the others into groups or families with similar chemical characteristics. Eighteen columns divide the elements into groups with closely. The periodic table arranges the elements, with their diverse physical and chemical properties, in order of atomic number and fits them into a logical pattern. Were it not for the simplification provided by this chart, students of chemistry would need to learn the properties of all 118 known elements. Periodic Table These are the 118 currently known and officially named elements that make up the periodic table (IUPAC 2016). Unfortunately for Meyer, his work wasn’t published until 1870, a year after Mendeleev’s periodic table had been published. The term “periodic” is based on the discovery that elements show patterns in their chemical properties at certain regular intervals. This 1868 table listed the elements in order of atomic weight, with elements with the same valency arranged in vertical lines, strikingly similar to Mendeleev’s table. Mendeleev left spaces for elements he expected to be discovered, and today’s periodic table contains 118 elements, starting with hydrogen and ending with oganesson, a chemical element first synthesized in 2002 at the Joint Institute for Nuclear Research (JINR) in Dubna, Russia, by a team of Russian and American scientists. The periodic table is an arrangement of the chemical elements, structured by their atomic number, electron configuration and recurring chemical properties.In the basic form, elements are presented in order of increasing atomic number, in the reading sequence. Its story is over 200 years old, and throughout its history, it has been a subject for debate, dispute and alteration.Īttempts to classify elements and group them in ways that explained their behavior date back to the 1700s, but the first actual periodic table is generally credited to Dmitri Ivanovich Mendeleev, a Russian chemist who in 1869 arranged 63 known elements according to their increasing atomic weight. Oganesson has several distinctions amongst all of the elements in the periodic table. Oganesson was discovered in 2002 and its properties defy our expectations based on trends in the periodic table. Go into any scientist’s office or lecture hall anywhere in the world and you are likely to see one. Element 118, the final element in our International Year of the Periodic Table series, is oganesson. There is no more enduring reflection of science than the Periodic Table of Chemical Elements, which sheds light not only on the essence of chemistry but physics and biology as well.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
